
In the first investigation of toxic fire retardants in parents and their children, Environmental Working Group (EWG) found that toddlers and preschoolers typically had 3 times as much of these hormone-disrupting chemicals in their blood as their mothers.
 The 20 children we tested had an average of 3.2 times more fire retardants polluting their blood than their mothers.
The 20 children we tested had an average of 3.2 times more fire retardants polluting their blood than their mothers.Laboratory tests – conducted for EWG by one of the world’s leading scientific authorities on fire retardants – found that in 19 of 20 U.S. families, concentrations of the chemicals known as PBDEs were significantly higher in 1.5- to 4-year-old children than their mothers. In total 11 different flame retardants were found in these children, and 86 percent of the time the chemicals were present at higher levels in the children than their mothers.
The tests also found a form of PBDEs known as Deca, a heavily used flame retardant that has largely escaped restrictions because few labs can reliably test for it. The tests showed Deca more often and in higher concentrations on average in children than their mothers. These high exposures early in life point to a previously undocumented, serious, and disproportionate risk to young children.
Eight of the 20 mothers we tested were also part of earlier EWG studies that found high levels of PBDEs in human breast milk and household dust. EWG tests of umbilical cord blood also found PBDEs in 10 of 10 newborns. The current study is the first to show that U.S. children have much higher levels of PBDEs in their blood than their parents and in fact bear some of the heaviest burdens of flame retardant pollution in the industrialized world.
 Kristi & Stella: "I chose not to learn the results of our tests because that would only lead to one thing – increased anxiety on my part. Instead, I chose to direct my energy on insisting that the government enact real protection for our children and ourselves. The burden of responsibility should not fall on my daughter's small shoulders, but on those of the manufacturers and legislators who have the power to protect her, and all of us."
Kristi & Stella: "I chose not to learn the results of our tests because that would only lead to one thing – increased anxiety on my part. Instead, I chose to direct my energy on insisting that the government enact real protection for our children and ourselves. The burden of responsibility should not fall on my daughter's small shoulders, but on those of the manufacturers and legislators who have the power to protect her, and all of us."PBDEs in everyday items like furniture, computers, televisions and other electronics migrate into the home environment and could expose children to concentrations exceeding the U.S. Environmental Protection Agency’s recommended safe level. Children ingest more PBDEs than adults because they stick to kids’ hands, toys or other objects they put in their mouths.
Children’s developing brains and reproductive systems are extraordinarily vulnerable to toxic chemicals. In the case of PBDEs, laboratory tests in peer-reviewed studies have found that a dose administered to mice on a single day when the brain is growing rapidly can cause permanent changes to behavior, including hyperactivity. Children's bodies may not metabolize and excrete toxic chemicals as readily as adults.
PBDEs, or polybrominated diphenyl ethers, are global pollutants that build up in the blood and tissues of people and other living things. Two forms of PBDEs known as Penta and Octa are no longer made in the U.S. because of health and safety concerns, but are still found in furniture and foam items made before the phase-out was complete. The largest volume of PBDEs are used in electronics in a form known as Deca. Deca is banned in European electronics and in some U.S. states.
The chemical industry is waging a high-stakes effort to keep Deca on the market, claiming it poses no health risk. But EWG’s tests show that Deca enters people’s bodies, and is polluting children’s blood at much higher levels than adults’. Deca was detected in 65 percent of children and 45 percent of adults.
 Laura Spark with twin daughters Naomi & Lea: Naomi had 6 times more PBDEs in her body than her mom did even though they live in the same home and eat the same foods.
Laura Spark with twin daughters Naomi & Lea: Naomi had 6 times more PBDEs in her body than her mom did even though they live in the same home and eat the same foods.Even as the chemical industry insists Deca is safe, manufacturers are moving away from the use of all chemical fire retardants and have found they can achieve fire safety through smarter product design. Major electronics manufacturers including Nokia, Sony-Ericsson and Samsung no longer use Deca and are phasing out other bromine-based fire retardants.
Despite the evidence that PBDEs are harmful, that they pollute people’s blood, and that safer alternatives are available, the EPA has done little to address children’s ongoing exposure. Deca remains widely used, and a regulatory loophole allows Penta, one of the PBDEs banned earlier, to enter the U.S. in imported furniture. Until Deca is banned in all consumer products, Penta is banned from imports, and fire safety regulations are revised to promote non-chemical solutions, American families – and especially their children – will continue to be needlessly exposed to these harmful compounds.
Study Participants
Environmental Working Group's study of fire retardants in toddlers and their mothers included 20 families from 16 cities in 11 U.S. states. Many of our study participants are pictured below.
Key:
Mother's PBDE level (ppb)
Child's PBDE level (ppb)
ppb = parts per billion in blood lipids
 |
 |
 |
||||
| 20 Moms [average PBDE level = 25 ppb] 20 children [average PBDE level = 62 ppb] children's ages = 1 to 4 |
Maija [63 ppb] & Eva [84 ppb] Taos, NM Attorney Eva's age = 2 |
Tess [12 ppb] & Nicolas [26 ppb] Portland, OR Lecturer in Microbiology Nicolas' age = 3 |
||||
 |
 |
 |
||||
| Laura [12 ppb] & Naomi [79 ppb] & Lea [not tested] Boston, MA Urban Planner Naomi's age = 4 |
Teri [16 ppb] & Natalie [25 ppb] San Francisco, CA Attorney Natalie's age = 3.5 |
Tracy [24 ppb] & Zade [82 ppb] Missoula, MT Registered Nurse Zade's age = 2 |
||||
 |
 |
 |
||||
| Kristi [results not disclosed] & Stella [results not disclosed] San Francisco, CA Director of Communications, Forest Ethics Stella's age = 2.5 |
Katrina [17 ppb] & Ruby [113 ppb] Oakland, CA Web Designer Ruby's age = 4 |
Greta [74 ppb] & Tavin [71 ppb] Seattle, WA Stay at Home Mom Tavin's age = 3.5 |
||||
 |
 |
 |
||||
| Bronwyn [13 ppb] & Teo [69 ppb] Riverside, CA Assistant Professor Teo's age = 2 |
Hillary [16 ppb] & Brynn [54 ppb] Colorado Springs, CO Lecturer Brynn's age = 4 |
Susan [17 ppb] & Louis [54 ppb] & Ella [not tested] Washington, DC Non-Profit Fundraiser Louis' age = 4 |
||||
 |
 |
 |
||||
| Laurie [22 ppb] & Conner [36 ppb] Missoula, MT University Faculty Conner's age = 3.5 |
Erika [19 ppb] & Hannelore [37 ppb] Seattle, WA Staff Scientist, Washington Toxics Coalition Hannelore's age = 3.5 |
Mary [31 ppb] & Olivia [70 ppb] Alameda, CA Co-Founder and Director, Making Our Milk Safe (MOMS) Olivia's age = 2 |
||||
 |
 |
|||||
| Jennifer [27 ppb] & Elijah [96 ppb] Helena, MT Attorney Elijah's age = 3 |
Liz [10 ppb] & Evan [34 ppb] Clinton, CT Pediatric Nurse Practitioner Evan's age = 2 |
|||||
Detailed Findings
 Nicolas had 2.2 times more PBDEs than his mom Tess.
Nicolas had 2.2 times more PBDEs than his mom Tess.
EWG examined concentrations of PBDEs in 20 American families. Paired tests taken on the same day from moms and kids found higher concentrations of toxic fire retardants in children from nearly every family. In 19 of 20 families, children had greater total PBDE measurement than their mothers, typically 3.2 times higher. The children studied ranged in age from 1.5 to 4 years old, a time when their playing and eating habits lead to increased ingestion of PBDEs and other contaminants that accumulate in the home. Total PBDE concentrations in children averaged 62 parts per billion in lipid, and ranged from 25 to 114 ppb (or 12.9 to 61.9 pmoles/gram lipid). Concentrations for mothers averaged 25 ppb, and ranged from 10 to 74 ppb (or 5.9 to 40.6 pmol/gram lipid).✳ The study suggests that U.S. children 1 to 4 years of age bear some of the heaviest burdens of flame retardant pollution in the industrialized world. Our study is the first systematic monitoring of Deca PBDE in children, which was identified as a critical data gap by EPA in 2005 (EPA 2005). We found 13 of 20 children and 9 of 20 mothers had measurable amounts of Deca PBDE—a widely used chemical that industry has argued poses no risk to health. Deca is found in plastic for TVs, computers and other electronics. However, the chemical is banned in Europe and some U.S. states, and most manufacturers are voluntarily replacing the chemical due to concerns about its toxicity, accumulation in people and breakdown into other toxic PBDEs.
 Olivia had 2.3 times more PBDEs than her mom Mary.
Olivia had 2.3 times more PBDEs than her mom Mary.
The finding of elevated PBDEs in young children is especially concerning because the most sensitive period for their toxic effects appears to be late pregnancy and early childhood. Single day exposures during a period of rapid brain development are linked to permanent changes in learning, memory and behavior. PBDEs are also known to disrupt thyroid and reproductive hormones, which could lead to permanent impacts if exposures occur during early life.
Children at risk
Our findings are strong evidence that children are not adequately protected from harmful contaminants. Efforts to document concentrations of pollution in people almost exclusively focus on healthy adults whose behaviors lead to lower exposures to many contaminants found in the indoor environment.
 Hannelore had 2 times more PBDEs than her mother Erika
Hannelore had 2 times more PBDEs than her mother Erika
However the handful of studies like ours, which focus on chemical exposures for young children, indicate that we can't adequately protect children without understanding how they differ from adults. A recent study examined concentrations of 52 industrial chemicals in 3 to 6 year old children, and found 20 chemicals were markedly elevated relative to older children (ages 12 to 18) and adults (Sexton 2006). These contaminants included volatile chemicals, PCBs, lead, mercury and persistent pesticides. Notably children’s exposures to many of these chemicals continue decades after the chemicals were banned from commerce. Childhood is a period of unique vulnerability to PBDEs and other toxic chemicals. The developing brain and reproductive systems are incredibly vulnerable to toxic chemicals. A classic example is childhood lead poisoning, which illustrates that children’s bodies may be permanently impacted by concentrations of a chemical that would not harm a healthy adult. In the case of PBDEs, a single exposure during a day of rapid brain growth and development can cause permanent changes to behavior and activity (Viberg 2003a, Viberg 2003b, Eriksson 2001, Viberg 2007). In contrast to the single day studies of PBDEs administered to mice in a controlled laboratory setting, American children are exposed to PBDEs thoughout pregnancy and early life, during many periods of significant brain development. They are also exposed to PCBs, lead, Teflon chemicals, and other industrial pollutants that affect brain and reproductive development.
Children in our study have consistently higher levels of PBDEs than adults
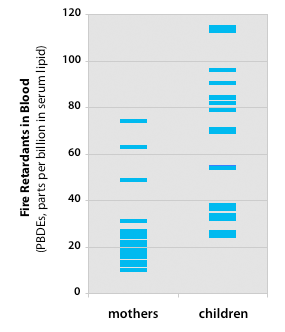
Source: EWG's tests of 20 children and mothers
In the case of lead poisoning, economists have quantified the societal impact of slight deficits in intelligence or behavior, which amount to billions of dollars of lost wages, special education, and other social services (Schwartz 1994). The decades of interventions to safely remove lead from children’s homes is being undermined by their exposure to new chemicals intentionally added to products for fire-resistance, or to make non-stick pans and non-stain fabrics, despite evidence they could pose similar risks to children’s wellbeing.
About PBDEs
PBDEs are a class of toxic fire retardants added to household furniture and electronic items. As persistent and bioaccumulative chemicals, their use has lead to accumulation in wildlife, food products, and the general environment. Two of 3 types of PBDEs were withdrawn from commerce in the U.S. in 2005, due to concerns of harmful effects to people and the environment. The third form, known as Deca, is still in widespread use. Studies indicate that daily exposures to PBDE can exceed the government’s safe daily exposure levels for some children. Duke University researchers calculated the amount of PBDEs sticking to adult and children’s hands and determined that young children were most at risk, due to normal play and exploration which leads to putting their hands and other non-food items in their mouths (Stapleton 2008). They estimate children's exposure to PBDEs in dust to be 10 times higher than that of adults.
 Maija's daughter Eva had 30% more PBDEs than she did
Maija's daughter Eva had 30% more PBDEs than she did
This is especially concerning because childhood is the period of greatest sensitivity to PBDEs' toxic effects. The most sensitive laboratory tests find that exposure to PBDEs during early life permanently affects learning, memory and behavior in juvenile animals (EPA 2008 a,b,c,d). EPA's recent safety value for Deca PBDE exposure is 500 times lower that the previous estimate by the National Academy of Sciences and promoted by the American Chemistry Council in 2003 (NAS 2000, ACC 2003). The reason is because EPA considered toxicity of Deca exposures during early childhood, while the NAS used a 1986 study that only considered toxicity in adult animals (EPA 2008d). Studies by EWG and others of umbilical cord blood, breast milk, childrens' blood and homes find that American children are exposed to these chemicals throughout early life, including during pregnancy, infancy and childhood (EWG 2003, 2004, 2005, Herbstman 2007, Wu 2007, Athanasiadou 2008).
 Zade had 3.4 times more PBDEs than mom Tracy
Zade had 3.4 times more PBDEs than mom Tracy
Tracy says: "My son's concentrations of PBDEs were 3.4 times higher than my own. While this study shows that is common for kids his age, it is certainly worrisome."
In the recent analysis of the health effects of the four most common PBDEs in laboratory studies, EPA determined that the most sensitive impacts are to mice brain and behavior after receiving a single dose of PBDEs (EPA 2008 a,b,c,d). The studies test a period of rapid brain development that is similar to brain growth during the 3rd trimester of a human pregnancy. But these tests do not account for the additional impact of exposures during other life stages, the cumulative impact of exposure to other PBDEs, and other toxic chemicals like mercury, lead and PCBs which also affect intelligence and behavior. As a result, the EPA admitted "low confidence" that the current “daily safe exposure level” for oral exposure assigned by the Agency fully protected health (EPA 2008 a,b,c,d). Scientists measuring PBDEs that accumulate on people's hands calculate that children ingest roughly 10 times more PBDEs than adults from hand-to-mouth contact (Stapleton 2008). Based on these estimates children (weighing 22 to 44 pounds) get about half their daily safe exposure to Penta type PBDEs from the chemical accumulating on their hands alone. The children with higher than average exposures would exceed the safety level based on hand-to-mouth exposures alone. In addition to dust ingestion children face PBDE exposure from foods, inhalation and directly mouthing items with PBDEs in them. Despite this worrisome evidence about PBDEs and the availability of safer replacements, U.S. health and environmental agencies have done little to address children’s on-going exposures to these chemicals. Deca continues to be widely used despite clear evidence that suitable replacements exist and a voluntary shift by major retailers away from the compound. Penta, the form used in foam furniture, is no longer produced in the U.S. However, gaps in EPA’s regulatory safety net allow Penta to legally enter the U.S. via imported foam products. Biomonitoring studies by CDC and others continue to focus on healthy adults and older children, overlooking the population most at risk.
Are current exposures safe?
 Susan's son Louis had 3.3 times more PBDEs than she did. Daughter Ella was not tested.
Susan's son Louis had 3.3 times more PBDEs than she did. Daughter Ella was not tested.
PBDEs have been proven to be especially toxic to the developing brain, leading to permanent changes in learning and behavior (EPA 2008 a,b,c,d). Laboratory studies administered single doses of each type of PBDEs to newborn mice and found permanent effects which worsen with age (Eriksson 2001, Viberg 2002, 2003a, 2003b, 2007). Similar findings have been confirmed by other scientists (Branchi 2003, Kuriyama 2005, Talsness 2005, Rice 2007). It is difficult to compare the doses administered in these 1-day studies to human exposures. PBDEs are widely detected in people, including nearly all of the 2,000 participants in a recent CDC study (Sjödin 2008). Body burden measurements indicate that Americans are exposed consistently from development in the womb through adulthood to a mix of PBDEs and other chemicals with similar impacts to neurodevelopment. A senior scientist at EPA estimates that PBDE concentrations in the most highly exposed Americans are within a factor of 10 from the doses found toxic in the laboratory (Birnbaum 2006). Many Americans are likely to exceed EPA's safety guideline, which calls for human exposures to be 300 to 3000 times lower than those found to be toxic in laboratory studies. Despite widespread exposure during pregnancy and early childhood, only a few studies have examined toxic effects of PBDE exposures in people. A case-control study in Denmark and Finland found statistically significant differences between PBDE concentrations in the breast milk of mothers of boys with cryptorchidism (in which one or both testicles is undescended at birth) as opposed to mothers with unaffected sons (Main 2007). The results from this study suggest that the mothers with higher body burdens of PBDEs were more likely to have sons born with cryptorchidism. However, placental levels of PBDEs did not correlate in the same manner as did the breast milk levels and it is not clear why. These findings are also difficult to interpret given that the concentrations in both groups studied are typically less than 5 parts per billion, dramatically lower than levels in Americans.
 Hillary's daughter Brynn had 3.4 times more PBDEs than she did.
Hillary's daughter Brynn had 3.4 times more PBDEs than she did.
A recent study from the U.S. looked at the effects of PBDE exposures on the hormone systems of a group of men who consume sport fish (Turyk 2008). The researchers found that in this group of men, exposure to PBDEs at levels similar to those found in the U.S. population were associated with significant effects on thyroid hormone levels. In addition, those men with the highest 5 percent of PBDE levels also had increased thyroid antibodies, which is a risk factor for thyroid disease later in life. Although this study did not include pregnant women, infants, or children, these results are especially worrisome because even minor fluctuations in thyroid hormone levels can have permanent negative effects on brain development and growth in the developing fetus, infants, and children.
PBDEs in young children
Ours is the first U.S. study to look carefully at PBDE levels in young children. The only previous U.S. study of children was of a single family with 2 young children living in Oakland, California (Fischer 2007). The study reported consistently high measurements of PBDEs and other chemicals in the toddler and 5 year-old child relative to their parents. The significant differences between children and their parents call into question the previous idea that concentrations in mothers is a good predictor of exposures to their children. While maternal monitoring is a proxy for exposure to the developing fetus or nursing infant, the concentrations we measured in young children were higher than levels reported in a large umbilical cord blood study for U.S. infants (Herbstman 2007). They are also higher than concentrations measured in teens and adults in CDC's recent study of 2,000 Americans. Interestingly the moms in our study had lower concentrations than the CDC group, which could be due to the limited number in our study. However, these findings point to the need to evaluate behavioral factors that lead to increased exposures during early life.
EWG findings indicate that young children have higher levels of PBDEs than other age groups
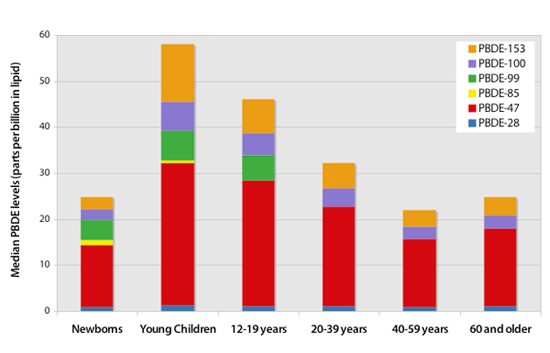
Graphic shows median values reported for 6 common PBDEs primarily used in the Penta mixture. Data are drawn from Herbstman's (2007) tests of 297 umbilical cord blood samples, EWG's test of 20 children ages 1 to 4 years old, and CDC's tests of 2,062 Americans age 12 and older (Sjödin 2008). Deca PBDE was not reported in newborn and adults, but estimated to be around 2 ppb in both groups.
Studies including young children in Norway and Australia both detected elevated concentrations in 0 to 4 year olds, relative to older children and adults (Thomsen 2002, Toms 2008). Notably, many studies of young children outside the U.S. find significantly lower PBDE levels. This is due to the fact that almost all of the Penta PBDE manufactured was used in the U.S. and thus resulted in the addition of thousands of pounds of toxic chemicals to home products. The average PBDE level for 0 to 4 year old Norwegian children was less than 10 ppb in lipid (Thomsen 2007). Each of our child participants exceeded this amount. Four-year-old Spanish children had an average of 1.3 to 3.6 ppb PBDEs in their serum, with higher concentrations noted in breastfed kids (Carrizo 2007). Another study examined paired samples from moms and kids in the Faroe Islands. In this study, children's concentrations at age 7 were roughly equal to maternal concentrations during pregnancy of 4 to 5 ppb in lipid (Fängstrom 2005). The Faroe Islander exposures to Penta-like PBDEs are likely limited to dietary sources. Deca concentrations are much more similar between Americans and Faroe Islanders. A study of 4 pooled blood samples collected from 0 to 4 year old Australian children found slightly higher concentrations than those measured in children in our study (Toms 2008a). Concentrations of PBDEs ranged from 63 to 80 ppb in lipid. The average age of children was 2 years. Each sample contained blood from 100 children. A follow up study was designed to take a closer look at the differences over the first 4 years (Toms 2008b). This study pooled samples from children 0 to 6 months in age, 6 to 12 months in age and 1, 2, and 3 year olds. This study found lower levels in the 0 to 6 month-old children, and similar concentrations for kids 6 months to 3 years. The authors concluded that increasing concentrations in children post-weaning suggest a role for interior sources of PBDEs. Notably the concentrations for Australian children were much lower in the second study, only 35 to 43 ppb (Toms 2008b).
Why do American children have such intense exposures?
 Laurie's son Conner had 60% more PBDEs than his mom.
Laurie's son Conner had 60% more PBDEs than his mom.
Childhood exposures to household items containing PBDEs have been implicated as the predominant route of daily exposure for U.S. children (Jones-Otazo 2005, Lorber 2008, Allen 2007, Stapleton 2005). This is because the US fire safety standards have been passed without in-depth consideration or any public discussion of likely chemical hazards, and thus resulted in thousands of pounds of toxic chemicals added to home products. Other countries don't require chemical fire retardants, and the U.S. accounted for roughly 95 percent of the world's annual Penta consumption before the mixture was phased-out. Moreover, as a result of widespread environmental contamination, PBDEs are now also detected in common foods, including meat, fish and dairy products (Schecter 2006), which are secondary sources of exposure. Researchers at Duke and Boston Universities have taken a close look at the role of household sources of PBDEs in the measurements in Americans. One recent study found a positive association between the concentrations of PBDEs in household dust and concentrations of PBDE in the bodies of nursing mothers (Wu 2007). Another took a detailed look at brominated chemicals in the home, and found a strong link between Deca measurements and televisions (Allen 2007) and found that PBDE levels in house dust did not vary significantly over an 8 month period (Allen 2008). Finally, researchers measured PBDE concentrations on the hands of adults and older children and used these numbers to estimate the daily intake for young children who have high rates of hand-to-mouth contact (Stapleton 2008). These findings indicate that PBDE particles stuck to hands contribute to about 50 percent of EPA's safe daily exposure level for 2 types of PBDEs for a typical small child, and highly-exposed children will exceed the safety level based on hand-to-mouth contact alone. Nursing can be a significant source of PBDEs for infants, but there is no indication from this or other studies that the harmful effects of PBDEs or other persistent contaminants outweigh the benefits of breastfeeding (Jorissen 2007, Dorea 2006). PBDE transfer from mother to child during pregnancy and nursing could skew the maternal-child comparison. However, studies of American mothers find that PBDE concentrations in mothers’ milk dropped by just 1 to 3 percent per month over the first 6 months of nursing when milk production is greatest (Hooper 2007). This is different than the observation that PCB levels slowly decline over the duration of nuring (LaKind 2001, Bloom 2007). The relatively steady concentrations of PBDEs is likely due to frequent and continuous contact with PBDE-containing consumer products, and possibly shorter half-lives than other persistent chemicals (Hooper 2007).
Benefits of breastfeeding outweigh contaminant concernsEWG strongly recommends that all mothers breastfeed their children if possible. The health benefits of breastfeeding are numerous and not overshadowed by concerns about contaminants in mothers' milk. For starters, breast milk contains essential fatty acids that are not present in infant formula. These components of milk may bolster brain and neurodevelopment to offset some of the impacts of chemicals like PBDEs (Jacobson 2002). Several studies find that in-utero, not breast milk, exposures to brominated fire retardants are more correlated with adverse health effects in children. These include findings of worse neurological outcomes and reduced growth based on in-utero not lactational PCB exposure (Jacobson, 2002, Koopman-Esseboom 1996), and lower body weight for height (Blanck 2002). Conversely studies of Michigan girls exposure to polybrominated biphenyls (PBBs) found that those with high pre- and post-natal PBB exposure underwent earlier menarche than girls with lesser exposures in-utero or who were not breastfed (Blanck 2000). Notably both PCBs and PBBs are structurally similar to PBDEs, and exhibit many of the same toxic effects. PBDEs have not been extensively tested in people. An additional group of studies find that breastfed children show lesser effects than formula-fed members of the cohort, despite higher body concentrations of toxic chemicals. These studies include those examining DDT, PCBs and dioxins as they affect infant neurodevelopment (Ribas-Fito 2007, Eskenazi 2006, Jorissen 2007, Jacobson 2002, Boersma 2000), mercury's effects on neurodevelopment (Jensen 2005) and DDT and asthma (Sunyer 2005, Sunyer 2006).More information about contaminants and breastfeeding |
 Liz's son Evan has 3.3 times more PBDEs in him than she does.
Liz's son Evan has 3.3 times more PBDEs in him than she does.
One Spanish study found higher concentrations of PBDEs in 4 year olds who had been breastfed (typically for about 4.5 months) compared to those who had not (Carrizo 2007). However, the absolute difference was small: an average of 3.6 ppb in the lipid of breastfed 4 year-olds compared to 1.3 ppb in formula-fed children. It is unclear how much of a role breastfeeding plays in our measurements. But U.S. children have both greater exposures to PBDEs in mothers' milk and in household products than children in other countries. The study of Australian children looked separately at 0 to 6 months and by 6 month to year increments until 4 years old. This study concluded that PBDE concentrations rise after weaning, so exposures in 2- and 3-year-olds were likely due to contact with household items (Toms 2008b). All children in our study were breastfed for at least 4 months, and many of them for much longer. We found no relationship between the duration of nursing and children's PBDE levels. Additionally 8 mothers and children from the group were participants in a 2003 study of PBDEs in breast milk. There was no clear relationship between a mothers' breast milk samples, her blood samples 3 to 4 years later, and her child's PBDE measurements at that age. In 5 cases children's blood concentrations were greater than PBDEs in mothers' milk. In the remaining 3 cases children's concentrations were lower. These findings underscore the importance of reducing PBDE exposures for women of childbearing age and children.
Types of PBDEs
 Jennifer's son Elijah had 3.6 times more PBDEs than his mom.
Jennifer's son Elijah had 3.6 times more PBDEs than his mom.
There are 209 types, or congeners, of PBDEs, and their name indicates the number and arrangement of bromine atoms in a molecule. Penta is the common name for a mixture of PBDEs used primarily in foam furniture. Penta mixtures vary, but typically include primarily PBDEs with 4 or 5 bromines, and a smaller amount of PBDEs with 6-bromines. The Octa mixture includes a mix of PBDEs with 6 to 10 bromines. The product known as Deca is primarily made up of PBDE-209 with 10 bromines. Deca formulations can include PBDEs with eight or nine bromines, higher purity formulations were recently introduced as a result of the European restrictions against PBDEs with 1 to 9 bromines. Overall, a small number of PBDE congeners predominate in commercial fire retardant mixtures, humans and in the environment. These include PBDE-47 (4 bromines), PBDE-99 (5 bromines), PBDE-153 (6 bromines), and PBDE-209 (10 bromines). Our tests revealed high concentrations of a trace PBDE in at least 4 participants. This is due to the fact that another brominated fire retardant, a polybrominated biphenyl (PBB-153) coeluted with PBDE-154 in our analysis.
✳ These sum values of PBDE do not include PBDE-154 which coeluted with a different brominated fire retardant PBB-153 in our analysis. This chemical was excluded in the total because at least 2 moms and their 2 children had abnormal amounts of this trace congener, suggesting relatively high levels of PBB-153. Hence our reported levels are slightly underestimating the total PBDE levels in the children and their mothers.
Kids and Contaminants
 Ruby had 6.6 times more PBDEs than were found in her mother Katrina
Ruby had 6.6 times more PBDEs than were found in her mother Katrina
"At first it was upsetting to know that Ruby had fairly high levels of PBDEs in her body. But it becomes even more upsetting when you think about the implications for all our children. There's nothing about our lifestyle that would put us at risk. If our levels are high, then yours probably are, too." Katrina Alcorn, study participant.
Children are particularly vulnerable to the toxic effects of harmful chemicals for a number of reasons. Their normal behaviors lead them to explore their local environment in ways that increase their exposures to toxic chemicals that may reside in everyday items like carpeting, furniture, electronic items and other common consumer products. Their small size leads to more food and water consumption, dermal exposure and higher inhalation rates than adults. They also have dietary patterns that differ greatly from adults, leading them to disproportionately high exposures from chemical contaminants in certain types of foods and beverages. Finally, their developing biological systems are not able to respond to environmental exposures in the same manner as adults, leading to a decreased ability to effectively detoxify once they have been exposed to toxic chemicals (Moya 2004). Public health protections for toxic chemicals often fail to consider these special factors and put our children at risk.
Children at play
 Greta and son Tavin had roughly equal concentrations of PBDEs.
Greta and son Tavin had roughly equal concentrations of PBDEs.
Children, as they are familiarizing themselves with their local environment, often handle everything around them and then place their hands into their mouths. This hand-to-mouth behavior is perfectly normal but places them at risk from chemical contaminants in soil, dust, and carpeting (Landrigan 1995). In addition, they handle toys and other objects and then lick their fingers or place these objects in their mouths, leading to ingestion of toxic chemicals that may reside in these products (Moya 2004). One study estimates that children ages 4 and younger put their hands in their mouths on average 16 to 18 times per hour (Tulve 2002). Children also play close to the ground, where they are exposed to chemical contaminants that find their way into grass and soil and that are found in dust that may coat indoor surfaces such as floors and carpets (Moya 2004). In general, these types of activities lead to exposure through ingestion and inhalation, but dermal exposures are also a concern, especially when it comes to soil contaminants, since soil can adhere to skin long after direct contact has ceased (Moya 2004).
Dietary Differences
Children eat more food and drink more water per unit of body weight than do adults (Landrigan and Carlson 1995). This means that kids have higher exposures to toxic chemicals that contaminate commonly consumed foods, beverages, and drinking water in comparison to adults (Landrigan and Carlson 1995). Children also often have a less varied diet than adults since kids often have specific food and beverage preferences and are not eager to try new foods. This means that children’s consumption of certain foods and beverages is disproportionately high in comparison with adults; for example, U.S. Department of Agriculture surveys have shown that kids consume more dairy products and produce items per unit of body weight than adults (Moya 2004). With respect to produce items, closer analysis has shown that young children eat more than twice the amount of common fruits and vegetables like carrots, broccoli, and grapes when compared with adults, leading to higher pesticide exposures among kids (NRC 1993). Children also consume more water per unit of body weight than adults do. Tap water testing by utilities across the United States has found hundreds of chemical contaminants, including pesticides, heavy metals, and endocrine disruptors (EWG 2005). Children, on average, drink from 2 to 5 times the amount of water per unit of body weight than do adults (Moya 2004). Babies who are fed powdered or concentrated liquid formula ingest large amounts of tap water over the course of their first year. These facts result in significantly higher exposures to tap water contaminants among children.
Biological Differences
The human body has certain biological mechanisms in place to metabolize and detoxify toxic chemicals. In children, these biological mechanisms are still developing and their immature organ systems do not have the same capacity as adults to metabolize and excrete toxic chemicals. For example, liver enzymes are critical in the process of detoxification; in children, these enzyme systems are not fully developed, leading to a decreased ability to effectively deal with toxic chemical exposures (EPA 2007).
 Bronwyn's son Teo had 5.1 times more PBDEs in him
Bronwyn's son Teo had 5.1 times more PBDEs in him
In addition, children’s organ systems are also rapidly developing during childhood; exposure to toxic chemicals during these periods of rapid development could lead to permanent and irreversible damage to these organ systems. This is especially true for the brain and nervous system, which are particularly vulnerable to toxic chemicals since they do not have good repair systems in place once structural damage has occurred. Exposure to nervous system toxins during childhood can decrease intelligence and cause behavioral problems. Lead poisoning is a classic example of a toxin that poses unique risks to children. The body's protective blood-brain barrier forms over the first 5 to 7 years. Prior to that, low level lead exposures can permanently damage the nervous system, lower IQ and cause behavioral challenges (EPA 2008e). Economists calculate that even the subtle deficits cause by low level lead poisoning tally up to billions of dollars in direct costs to society. These include lost wages, social services, and educational interventions. Clearly these findings should lead us to invest up-front in assessing new chemicals for their ability to harm children's development. While ingestion of toxic chemicals is the most significant route of exposure for children, dermal and inhalation exposures are important considerations with certain chemicals like pesticides. Children play close to the ground in activities that involve contact with soil and dust. Kids also have higher surface area when compared with adults so dermal exposures can be more significant in children (EPA 2007). As a result, the potential dose of a chemical following dermal exposure is estimated to be about 40 percent greater in children than adults. Children have higher rates of inhalation than adults because of higher metabolic rates, leading to greater exposure to toxic air pollutants. Children’s unique behaviors and physiology make them particularly vulnerable to environmental exposures. Once these exposures have occurred, their immature organ systems may become irreversibly damaged and unable to detoxify and excrete toxic chemicals as effectively as adults. Unfortunately, most state and national biomonitoring programs do not include young children, leaving huge deficiencies in our understanding of how toxic chemicals may affect them. These deficiencies must be adequately addressed when lawmakers and scientists propose safety standards for chemicals because it is clear that standards that protect adults may not necessarily protect children.
Children's diseases on the rise
 Teri's daughter Natalie had 60% more PBDEs than her mother.
Teri's daughter Natalie had 60% more PBDEs than her mother.
Recent U.S. health trends among children raise concerns about the role of environmental exposures in increasing rates of certain chronic diseases. Asthma rates doubled from 3.6 to 7.5 percent from 1980 to 1995, and these rates have continued to increase. The EPA estimates that 9.3 percent of children suffered from asthma in 2006 (EPA 2008f). Neurodevelopmental disorders among children have also increased in recent years, with autism now affecting 1 in 150 children and ADHD diagnosed in 1 of every 15 children ages 5 to 17. Cancer in children increased by 33 percent from 1975 to 2005 but thankfully, mortality rates decreased by almost half during that time period due to better treatment options (EPA 2008f). Acute lymphocytic leukemia and brain cancer incidences have particularly seen increases among children from the 1970’s to the 1990’s. These types of increases in chronic diseases among children are worrisome but unfortunately not unexpected given their unique vulnerabilities to environmental exposures and the overabundance of toxic chemicals in our air, tap water, food, and local environments. The U.S. needs effective national chemical policies that take into account children’s unique behaviors, diets, and biology in order to minimize the effects of toxic chemicals on their health.
Deca Risks
This study presents evidence that U.S. children are widely exposed to Deca PBDE. Deca was measured in more than half of our participants, with higher levels in children. Median concentrations in children were 1.74 ppb. Average levels in children with detectable Deca was 4.7 ppb, with 2 children between 10 and 20 ppb lipid. Deca made up about 20 percent of the total PBDE measurements in 6 of our participants when congeners were evaluated on a molar basis. As with the other PBDEs children had higher concentrations than adults.
Deca is the most widely used PBDE, and the only form still produced in the United States. Yet Deca has evaded regulation due to the fact that it is not commonly measured in biomonitoring studies. Few laboratories have the capability to reliably detect it, due to its chemical structure and because it is a common contaminant in laboratory equipment which often complicates the quantification of low parts per billion concentrations.
Deca also passes through the body quickly, with an estimated half life of just 2 weeks, as opposed to other PBDEs with longer half-lives (Thuresson 2005). The short-residency time complicates biomonitoring studies because 75 percent of a single day exposure will be gone one month later. The finding of this chemical in more than half of participants indicates frequent ingestion of Deca via contaminated dust or foods.
We found higher Deca concentrations in kids than their mothers
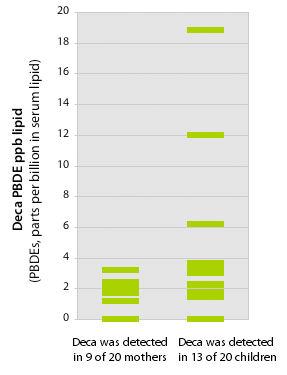
Source: EWG's tests of 20 children and mothers
Like other PBDEs, Deca upsets the developing brain and reproductive system (EPA 2008d). But recent studies indicate that it also impacts the reproductive system, possibly at even lower exposure levels (Van der Ven 2008). In addition to its direct toxicity there are serious concerns that Deca breaks down in the environment to form PBDEs with fewer bromines which are more persistent and bioaccumulative in people. Deca is quickly transformed in humans even though it is still unclear into exactly which breakdown products. In workers exposed to Deca there was a clear accumulation of PBDEs with 8 or 9 bromines (Thuresson 2005). Similar results were found in rainbow trout and rats fed Deca in controlled studies (Kierkegaard 1999, Huwe 2007).
Deca manufacturers have claimed that the chemical does not pose a health risk because it is poorly absorbed, quickly excreted and therefore much less of a health concern than other PBDEs (ACC 2003). They also dismiss concerns that Deca breaks down in the environment to form Penta-type PBDEs (BSEF 2006). But these claims have been disputed by dozens of scientific publications and recent reviews by environmental and health agencies in the European Union, Canada and U.S. States of Maine, Michigan, Illinois and Washington.
Minnesota's Pollution Control Agency recently concluded that Deca shows potential to accumulate and concentrate in the food chain, is toxic, and breaks down in the environment to more harmful forms. They also found that effective alternatives are available (Minnesota PCA 2008). The Canadian government echoes these findings (Canada 2006). Michigan's 2008 summary and Washington State's 2006 review both find similar risks of harm and support a legislative ban on Deca-BDE contingent on the availability of a safe alternative (Michigan 2008, Washington 2006). The State of Maine also concurred with findings of Deca toxicity and breakdown and concludes that safer alternatives exist (Maine 2007). In 2007 Maine became the first U.S. state to fully ban Deca in its major uses.
What do biomonitoring studies tell us about Deca risks?
The measurements for young children indicate that Deca may pose a greater health threat than previously thought. Concentrations in 1/3rd of our child participants exceeded the CDC’s estimate of about 2 ppb for adults and older children. This estimate is calculated from pooled blood samples that do not give information about highly exposed sub-populations (Sjödin 2008).
The highest concentrations of Deca in our children were 12 and 19 ppb lipid, and were comparable to the only other tests of young children in the U.S., which was 2 siblings in California. The California study reported concentrations of 9 and 12 ppb in the older sibling, and 19 and 26 ppb in the younger, though much higher concentrations were reported in analysis by a different laboratory for samples taken 3 months prior (roughly 10 times higher in every family member) (Fischer 2006). Since Deca has a rapid metabolism in people, concentrations could vary widely within the 3 month sampling period. Like our test subjects these children's parents did not report any unusual sources of exposure.
Deca PBDE has now been reported in human blood from residents of Sweden, Nicaragua, Norway, the Faroe Islands, Belgium, the United Kingdom and Mexico (as reviewed by Athanasiadou 2008). Toxic metabolites of Deca, including hydroxylated PBDEs, are also detected as evidence of recent Deca exposure (Athanasiadou 2008). It appears to be detected more in umbilical cord blood, breast milk, and placenta than other body fluids (Frederiksen 2008, Gomara 2007).
Recent detections in wildlife, specifically top predators including grizzly bears and hawks, raise the issue of biomagnification in the food chain and additional concerns about the safety of this chemical for wildlife and ecological systems (Voorspoels 2006, Voorspoels 2007, Christensen 2005).
Deca breakdown
Deca breakdown has been documented in the bodies of fish and mice in laboratories and in wild animals (Stapleton 2004 Huwe 2007, Kierkegaard 1999, Van den Steen 2007). It also occurs in the environment by sunlight or bacteria, and has been documented in plastics, household dust, wastewater, and soil (Ahn 2006, He 2006, La Guardia 2007, Stapleton 2007, Kajiwara 2008).
We detected PBDEs with 7 and 8 bromines which are not commonly reported in U.S. studies and could indicate Deca breakdown or contact with items containing the seldom used Octa PBDE mixture.
PBDE-183, a type with 7 bromines, was found 6 children (30%) and 4 mothers (20%). All but one of these participants had detectable Deca, and the highest concentrations of BDE-183 was measured in the child with the highest Deca level. BDE-183 is not in the Deca mixture but would be an expected breakdown product. It is also a major component of the less common Octa mixture (La Guardia 2006).
Another trace congener, PBDE-197, containing 8 bromines was detected in every participant. Maximum concentrations were 2 ppb in lipid. PBDE-197 has been reported as part of the Octa mixture (LaGuardia 2006). Additionally rodents fed Deca had 10-fold increase of PBDE-197 in their bodies, which authors concluded was due to debromination (Huwe 2007). PBDE-197 and other PBDEs with 8 or 9 bromines have been detected in Deca-spiked dust exposed to sunlight, indicating debromination under real-world conditions (Stapleton 2007). PBDE-197 and Deca have been reported at elevated levels in the serum of Chinese electronics dismantlers (median of 310 and 83.5 ppb and a maximum of >3,000 ppb) (Qu 2007, Bi 2007). Scientists conclude that the level of these trace congeners is too high to be from impurities in the Deca mixture (Stapleton 2006).
Fire Retardants (PBDEs) in Toddlers: How to Avoid PBDes
There is growing awareness that current laws fail to protect children from harmful chemicals. PBDEs are just another example of the fact that policymakers allow calculated risks that many parents would prefer to avoid when it comes to something as critical and as fragile as their child's health. We urge you to support our efforts to reform the nation's safety net, and force chemical makers to assure that their products are safe for children before they are allowed on the market.
"This study felt like a real wake up call to me. I had never heard of PBDEs before participating in the study. It never occurred to me that chemicals inside the couch, mattress, pillow, or car seat could be harmful. And I didn’t realize that chemicals could be put in products with no proof of safety." Laura Spark, mom and kids study participant.
Children's susceptibility to chemicals is not limited to PBDE exposures. You can take simple steps to reduce your children's contact with harmful chemicals that are found in our homes, foods, and other products we use daily. These include chemicals found in body care products, non-stick pans, plastics and food itself. We also have tips for bolstering your and your kids' bodies against chemicals' effects. Download 10 Tips for a Healthy Home: A Parent's Guide to Going Green.
Avoid PBDEs and other harmful fire retardants in electronics
PBDEs are widely used in electronic items, and residues are detected in nearly every household sampled. Home tests show that common culprits are televisions, stereo or entertainment systems, power strips, and routers. Lesser concentrations are found in speakers, alarm clocks, phones, and device chargers (Allen 2007). Fortunately many manufacturers are shifting away from Deca due to restrictions in Europe and actions on the part of some U.S. states. In fact, Washington State estimated that about 57% of televisions and 95% of computer products do not contain PBDEs (Washington State 2006). When purchasing new products look for these brands, which have publicly committed to phasing out all brominated fire retardants: Acer, Apple, Eizo Nanao, LG Electronics, Lenovo, Matsushita, Microsoft, Nokia, Phillips, Samsung, Sharp, Sony-Ericsson, and Toshiba Panasonic has agreed to eliminate all bromine-containing fire retardants from mobile phones and computers by 2011, but does not give a commitment or timeline for the remainder of their products. Dell has incomplete restrictions. Motorola's phase-out of BFRs is limited to their ECOMOTO phone line. The following companies have or are phasing out Deca, but may use other bromine-based fire retardants in their products: Canon, Daikin, Intel, IBM, HP (Hewlett Packard), Minolta, Mitsubishi, Motorola, NEC, Nokia, Xerox. Scan your house for Deca-containing items. The chemical can be found in:
- Electronics TV components, mobile phones, fax machines, remote controls, video equipment, printers, photocopiers, toner cartridges, scanners.
- Transportation electronic components, automobile fabrics, plastics and electronics.
- Household items kitchen appliances, fans, heaters or hair dryers, curtains and drapes, water heaters, and lamp sockets.
Avoid PBDEs in foam
PBDEs were widely used in older foam furniture. The Penta form used in foam was not produced in the U.S. after 2004, but stockpiles of the chemical could have been used later (Betts 2008a), and the chemical could be present in imported items. Foam products that might contain these PBDEs include: couches, upholstered chairs, mattresses, futons, foam carpet padding, children's car seats, automobile interiors, foam pillows (including breastfeeding pillows), and other foam items. For many families replacing these items is not a possibility. You can still take simple steps to reduce your family's contact with these chemicals. 1. Inspect foam items. Replace anything with a ripped cover or foam that is misshapen and breaking down. If you cannot replace these items try to keep the covers intact. Beware of older items like car seats where the foam is not completely encased in a protective fabric. 2. Use a vacuum fitted with a HEPA filter. These vaccuums are more efficient at trapping small particles and will likely remove more contaminants and other allergens from your home. HEPA-filter air cleaners may also reduce particle-bound contaminants in your house. 3. Do not reupholster foam furniture. Even those items without PBDEs might contain poorly studied fire retardants with potentially harmful effects. 4. Be careful when removing old carpet. The padding may contain PBDEs. Keep your work area isolated from the rest of your home. Clean up with a HEPA-filter vacuum and mop to pick up as many of the small particles as possible. 5. When purchasing new products ask the manufacturers what type of fire retardants they use. Avoid products with brominated fire retardants, and opt for less flammable fabrics and materials, like leather, wool and cotton. Be aware that "natural" or latex foam will also contain fire retardants. SOURCES: Betts 2008a, BSEF 2006, Curtis 2007, Greenpeace 2008, Washington State Department of Ecology and Department of Health 2006.
Gov't and Industry Actions to Phase Out PBDEs
The widespread contamination by PBDEs of people, wildlife and the environment is the result of weak environmental laws that allow poorly tested chemicals to be used in consumer products, and a system that is slow to react to evidence that a chemical is accumulating in people. Swedish researchers first discovered PBDEs in samples of mothers' milk in 1999. The discovery was purely accidental, but sleuthing and further testing of stored breast milk samples from the 1970's lead the chemists to the startling finding that concentrations in breast milk had been doubling every 5 years since 1972 (Hooper 2000). In retrospect it seems somewhat obvious that these chemicals would be of concern. PBDEs are closely related to other persistant pollutants, polychlorinated biphenyls (PCBs) and polybrominated biphenyls (PBBs), which were both banned after researchers discovered that they accumulate in people and wildlife and are toxic. The Swedish government was among the first to ban each of the 3 PBDEs, and scientists note a resulting decrease in levels of these toxic chemicals in their population. The European Union has followed suit. However, U.S. policies leave large loopholes for continued exposure to PBDEs and other harmful fire retardants. Penta and Octa are not manufactured here, but can enter the U.S. in imported products. The safety of Penta replacements has been called into question, and there are no Federal restrictions on Deca use. Facing Federal inaction 11 U.S. states have passed bans, and 4 additional states have proposed or are considering legislation that would ban these chemicals.
International action on PBDEs
Fire retardants are not widely required in foam furnishings outside the U.S. Instead, less flammable materials and control of cigarettes and other ignition sources are used to assure fire safety. As a result Penta PBDE was almost exclusively used in the United States, and residents of other industrialized nations have much lower concentrations of these PBDEs in their bodies. The European Union has been more proactive in dealing with PBDEs than the U.S. Germany, Sweden and the Netherlands banned Penta PBDEs in the late 1980s and early 1990s. The E.U. banned the use and marketing of Penta and Octa PBDE’s in 2003 after evidence of a worrisome increase in PBDE levels in nursing mothers (Meironyte 1999, Noren 2000). Interestingly, PBDE levels in Swedish women appear to have declined significantly since the ban was enacted (Fängstrom 2008). While these regulations also initially included Deca, industry challenged the Deca regulations in court. The highest court in the E.U. recently upheld the ban of Deca in electronics, to take effect July 1st of this year (Betts 2008b). Both Sweden and Norway had also taken actions independently on Deca, with Sweden banning use of Deca starting January 2007 and Norway banning use starting in April 2008. The Canadian government has categorized PBDEs as “toxic” but has yet to take any regulatory actions.
U.S. action on PBDEs
Federal action: Penta and Octa PBDEs are no longer produced in the U.S. but regulatory loopholes provide incomplete protection from these and other toxic fire retardants. In 2004 the Great Lakes Chemical Corporation, the only U.S. manufacturer of Penta and Octa announced they would stop manufacturing the chemical as of January 2005. It is unclear when the final stores of this chemical were depleted. EPA subsequently passed a Significant New Use Rule that disallows Penta and Octa production but the chemical appears to be produced in China, and importing Penta-containing items is still legal. Penta in foam was replaced with newer bromine-containing fire retardants, and other poorly studied chemicals, including Tris--a chemical banned from use in children's sleepware in the 1970s. Penta manufacturers now sell an alternative, bromine-based fire retardant known as Firemaster 550. This product contains poorly studied chemicals that have recently been detected in household dust and sewage sludge (Betts 2008c). There are no restrictions on Deca use at the Federal level.
State Deca bans (enacted)
In the face of Federal inaction many state legislatures have placed restrictions on these chemicals (Curtis 2007, National Caucus of Environmental Legislators 2008). * Maine: Bans use of deca in furniture and mattresses effective as of January, 2008 and use of deca in electronics to take effect in 2010. * Washington: Bans use of deca in mattresses starting in 2008 and in furniture and electronics to take effect by 2011 provided safer alternatives can be found.
State Deca bans (proposed)
* Alaska (HB 271)- proposed ban on sale of items that contain Penta- and Octa-PBDEs and proposed ban on Deca in mattresses and electronics. Current status: Active. Bill Information * California (AB 706) - proposed ban on brominated and chlorinated fire retardants in furniture and bedding to take effect in January, 2010. Current status: Failed to pass the Senate 8/26/08. Bill Information * Connecticut (HB 5805) - proposed ban on Penta- and Octa-PBDE to take effect on July 1, 2008 and proposed ban on Deca in mattresses to take effect in 2010 and in electronics to take effect in 2011. Current status: Active. Bill Information * Hawaii (HB 461)- proposed ban on use of Deca in mattresses, furniture, and electronics to take effect July 2010. Current status: inactive. Bill Information * Illinois (HB 5298)- the state's Environmental Protection Agency has reviewed evidence and recommended that the Governor support a managed state-level phase-out of several DecaBDE’s uses. A proposal to prohibit Deca use in mattresses and furniture, or any other textile product intended for indoor residential use starting in January, 2008 and in electronics beginning in 2011. Current status: active. Bill Information * Maryland (HB 1)- proposed ban on Deca-PBDE to take effect in 2010. Current status: inactive, (failed to pass Senate Committee, April 2008). Bill Information * Michigan (HB 4465)- the state's Department of Environmental Quality recently reviewed Deca toxicity and decided the evidence was sufficient to support a legislative ban on Deca provided that safe alternatives can be found (Michigan DEQ 2008). Proposed ban on Deca-PBDE in furniture and mattresses to take effect in 2008 and in electronics in 2011. Current status: active. Bill Information * Minnesota (SF 651)- proposed ban on Deca in mattresses, furniture, and electronics to take effect July, 2010. Current status: inactive (vetoed by Governor Pawlenty in May, 2008). Bill Information * New York (A 07977, S 05244)- proposed ban on Deca-PBDE in furniture and mattresses to take effect July, 2010. Current status: active.Bill Information * Vermont (H.589, S.260) proposed ban on Deca in mattresses and furniture by 2009 and in electronics by 2011. Current status: active. Bill Information
State Penta and Octa bans enacted
* California: Bans use of Penta and Octa effective as of January, 2006. * Hawaii: Bans use of Penta and Octa effective as of January, 2006. * Illinois: Bans use of Penta and Octa effective as of January, 2006. * Maine: Bans use of Penta and Octa effective as of June, 2006. * Maryland: Bans use of Penta and Octa effective as of October, 2008. * Michigan: Bans use of Penta and Octa effective as of June, 2006. * Minnesota: Bans ue of Penta and Octa effective as of January, 2008. * New York: Bans use of Penta and Octa effective as of January, 2006. * Oregon: Bans use of Penta and Octa effective as of January, 2006. * Rhode Island: Bans use of Penta and Octa effective as of January, 2006. * Washington: Bans use of Penta and Octa effective as of January, 2008.
State penta and octa bans (proposed)
*Alaska (HB 271): proposed ban on sale and construction of products that contain penta and octa. Current status: active. More information at: http://www.legis.state.ak.us/basis/get_bill.asp?bill=HB%20271&session=25 * Connecticut (HB 5805): proposed ban on use of penta- and octa-PBDE to take effect on July 1, 2008. Current status: active. More information at: http://www.cga.ct.gov/asp/cgabillstatus/cgabillstatus.asp?selBillType=Bill&bill_num=5805&which_year=2008 Sources: McDonald 2005, Betts 2008c, Environment California 2004, National Caucus of Environmental Legislators 2008 (3/17/08 version), Curtis 2007, Illinois EPA 2007, Maine Department of Environmental Protection 2007, Michigan Department of Environmental Quality 2008.
Appendix: Study Details
About Participants
Paired blood samples were collected on the same day from 20 mothers and their first born children. Our child participants were 1.5 to 4 years old (average age 3 years). All were were living in the same house as their mother during the study. 8 of 20 mothers were multiparous, and 3 more were pregnant at the time of blood testing. The majority of our study participants are Caucasian. 1 participant is African-American and another is Latina. Their ages range from 28 to 44 years old. All are college graduates. All children were breastfed as infants for at least 4 months, with 14 of 20 for longer than 1 year. This is a longer duration of breastfeeding than is typical for American children. The study participants reside in 16 cities in 11 U.S. states: Alaska, California (5), Colorado, Connecticut, the District of Columbia (3), Massachusetts, Minnesota, Montana (3), New Mexico, Oregon, and Washington (2). We collected substantial information from participants about their diet, household products, occupation and places of residence. We found no obvious relationship between PBDE concentrations and mother or child's age, mother's occupation, duration of nursing, participant's body mass, diet or other household factors. None of our participants reported unusual contact with foam furnishings or electronics in their home, daycare or workplace.
Study process
All adult participants gave informed consent prior to study enrollment with both custodial parents giving consent for children's participation. The study received Institutional Review Board approval (Independent Review Consulting, Inc., Corte Madera, CA, USA). The blood sampling was performed between September 2006 and January 2007, at 16 commercial laboratories. Laboratory technicians centrifuged the samples and pipetted the serum into vials, using chemically clean glassware provided by EWG. Phlebotomists were instructed to fill a 10 ml vaccutainer as completely as possible from the child, and then collect 2 similar-size samples from the mother. This sampling design sought to both maximize serum available from young children, and to minimize any chance that larger sample volume for mothers would result in systematic differences between mother and child measurements. Two equal volume samples were collected from mothers and both samples were analyzed for QA/QC purposes. The samples contained an average of 3.5 grams of serum for children and 3.4 grams for adults. The chemical analyses were performed by Dr. Åke Bergman and colleagues at Stockholm University's Department of Environmental Chemistry. This is a highly regarded laboratory with decades of publications on PBDE and PCB levels in biological and environmental samples. The laboratory used GC/MS, using authentic reference substances. Samples were analyzed for 20 PBDE congeners and 11 were detected, each in at least 25% of participants. Congeners detected include: PBDE-28, -47, -85, -99, -100, -138, -153, -154/BB-153, -183, -197, -209. The following congeners were not detected: PBDE-17, -66, -71, -196, -201, -203, -206, -207, -208. We collected 40 primary samples (20 maternal, 20 children), as well as double samples from 17 of 20 mothers. There was high concordance between measurements for the 2 maternal serum samples as well as the 10 split samples performed by Stockholm University. All results were background adjusted and reported as concentrations in lipid. When duplicate samples were analyzed we averaged the 2 reported values.
Appendix: Detailed Results
| Children (N=20) | |||||
| Congener | median ng/g lipid | median pmol/g lw | min ng/g lipid | max ng/g lipid | percent quantified |
| BDE-28 | 1.0 | 0.4 | 0.3 | 2.2 | 100% |
| BDE-47 | 30.6 | 14.9 | 11.5 | 65.4 | 100% |
| BDE-85 | 0.4 | 0.2 | ND | 2.2 | 75% |
| BDE-99 | 6.2 | 3.5 | 1.8 | 15.5 | 100% |
| BDE-100 | 6.2 | 3.5 | 2.1 | 13.6 | 100% |
| BDE-138 | ND | ND | ND | 0.3 | 35% |
| BDE-153 | 12.5 | 8.1 | 3.4 | 31.9 | 100% |
| BDE-154/BB-153 | 3.2 | 2.0 | 0.4 | 38.0 | 100% |
| BDE-183 | ND | ND | ND | 1.7 | 30% |
| BDE-197 | 0.5 | 0.4 | 0.1 | 2.0 | 100% |
| BDE-209 | 1.7 | 1.7 | ND | 18.8 | 65% |
| Sum of 10 congeners# | 69.3 | 39.0 | 24.5 | 114.5 | 100% |
| Mothers (N=20) | |||||
| Congener | median ng/g lipid | median pmol/g lw | min ng/g lipid | max ng/g lipid | percent quantified |
| BDE-28 | 0.4 | 0.2 | 0.2 | 2.5 | 100% |
| BDE-47 | 8.8 | 4.3 | 3.1 | 39.9 | 100% |
| BDE-85 | ND | ND | ND | 0.4 | 10% |
| BDE-99 | 1.5 | 0.8 | 0.4 | 5.2 | 100% |
| BDE-100 | 1.2 | 0.7 | 0.4 | 7.0 | 100% |
| BDE-138 | ND | ND | ND | 0.5 | 5% |
| BDE-153 | 5.8 | 3.7 | 1.4 | 32.5 | 100% |
| BDE-154/BB-153 | 1.8 | 1.1 | 0.7 | 41.3 | 100% |
| BDE-183 | ND | ND | ND | 0.5 | 25% |
| BDE-197 | 0.3 | 0.2 | 0.1 | 0.8 | 100% |
| BDE-209 | ND | ND | ND | 3.2 | 45% |
| Sum of 10 congeners# | 18.2 | 10.5 | 10.1 | 73.8 | 100% |
Congeners not detected in any sample: BDE-17, BDE-66, BDE-71, BDE-196, BDE-201, BDE-203, BDE-206, BDE-207, BDE-208
# BDE-154 was not included in the summary value due to co-elution with PBB-153.
Acknowledgements
Special thanks to each of the 20 women and their children who volunteered to participate in this study. The donation of your time and blood samples will help to further our understanding about children's exposures to toxic fire retardants. The information from this study will add to the growing body of science that shows that children have unique exposures and vulnerabilities to toxic chemicals and will help to drive national chemicals policy reform that protects our most vulnerable populations.
We are also grateful to Dr. Åke Bergman and colleagues at Stockholm University for analyzing the samples and providing invaluable assistance and guidance throughout this study.
Thanks to EWG interns Rachel Gelbmann, Stephanie Graeter, Maya Kelmelis, and Dan Myers who assisted with research for this report. Thanks also to advocacy groups who helped us recruit study participants, Alliance for a Healthy Tomorrow, Institute for Agriculture and Trade Policy, and Washington Toxics Coalition.


